Valence and Conduction band are the two different energy levels separated by a certain amount of energy. The main difference between the valence band and conduction band is that valence band specifies the energy level of electrons present in the valence shell of an atomic structure. As against a conduction band holds those electrons that are responsible for conduction.
Let us now see on what factors the two bands are differentiated using comparison chart.
Content: Valence Band Vs Conduction Band
| Basis for Comparison | Valence Band | Conduction Band |
|---|---|---|
| Abbreviated as | VB | CB |
| Existence wrt fermi-level | It is present below fermi-level. | Its existence is above fermi-level. |
| Effect of external excitation | Electrons move out of valence band | Electrons reaches the conduction band. |
| Energy state | Lower | Comparitively higher |
| Electron density | High | Low |
| Force by nucleus | Strong | Weak |
| Presence of electrons causes band to be | Partially or completely filled. | Empty or partially filled. |
Definition of Valence Band
It is basically defined as the energy band that comprises of valence electrons present in the outermost shell of an atomic structure. These valence electrons, when provided with sufficient energy, get changed into free electrons and moves to conduction band thereby causing conductivity.
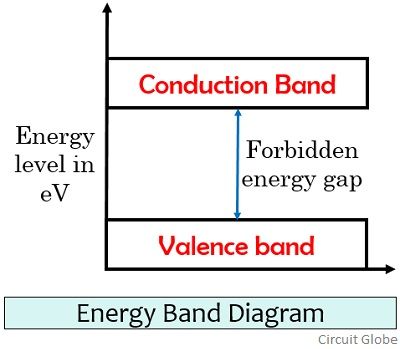
It is sometimes abbreviated as VB and is at a lower energy level than the conduction band in the energy level diagram. These two bands are separated by a certain amount of energy known as the forbidden energy gap. This energy gap depends on the type of material i.e., either the material is conductor, insulator or semiconductor.
As this band is a lower energy state, therefore, by the application of externally applied potential, the electrons in this band moves out of it and transfer towards the higher state so as to allow conduction through the material.
Definition of Conduction Band
A conduction band is defined as that energy band that consists of free electrons that are responsible for conduction. Thus this band is named so. Basically the electrons that get moved out from the valence band by experiencing external force reaches the higher energy band in order to support conduction.
This band is present above the fermi energy level i.e., is at higher energy state. Thus, electrons require a large amount of excitation energy to reach conduction band thereby giving rise to electric current. It is abbreviated as CB and it allows free movement of electrons inside it.
As we have already discussed that the forbidden energy gap between valence and conduction band is different for different material.
Like in case of conductors the two bands overlap and thus the electrons present in the lower energy band can easily move to the conduction band. But in the case of semiconductors, a sufficient energy gap exists between the two bands, therefore, external energy is needed to free the valence electrons. While for insulators the bandgap is very large and thus extremely large energy is required to make the valence electron a free electron.
Key Differences Between Valence Band and Conduction Band
- Valence band is present below the fermi energy level. While the conduction band is present above the Fermi level in the energy band diagram.
- When external excitation is provided then electrons present in the valence band moves out of it. However, because of external energy, the electrons move into the conduction band.
- The density of electrons is comparatively higher in valence than in conduction band.
- At room temperature, the valence band is usually either partially or completely filled. Whereas the conduction band is generally empty or partially filled at room temperature.
- Valence band is a lower energy state, whereas the conduction band is comparatively at a higher energy level.
- The electrons in valence band experiences strong force by the nucleus. As against the nucleus exhibits very weak or almost negligible force on the electrons at the conduction band.
Conclusion
So by this discussion, we can say that various factors exist that differentiates valence from the conduction band. But most importantly the two are differentiated on the basis of their presence in the energy band diagram.

Good explanation, but a doubt
Why valence electrons are moved freely and take part in electric conduction?
It is already explained in the content. You can find the answer to your question in the first paragraph of the definition of the valence band itself.